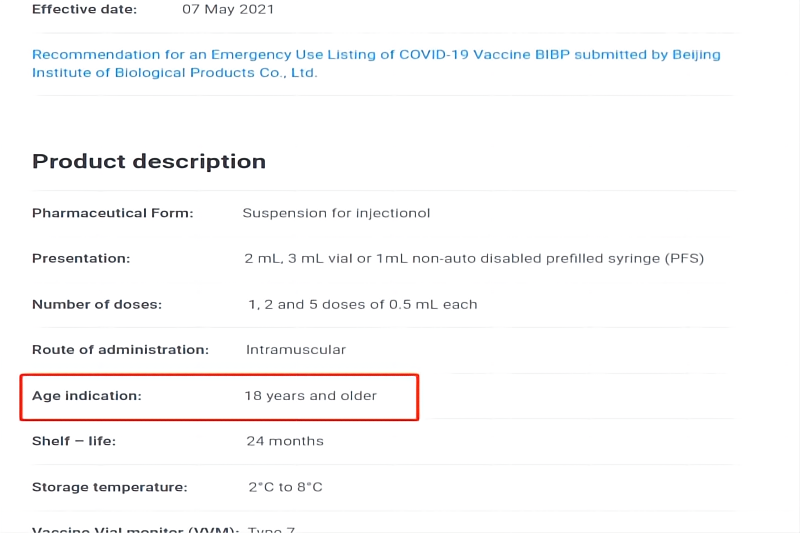
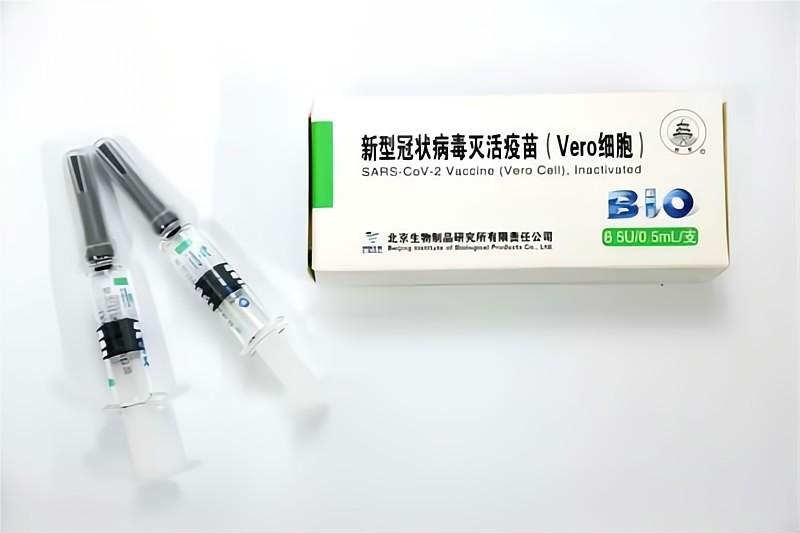
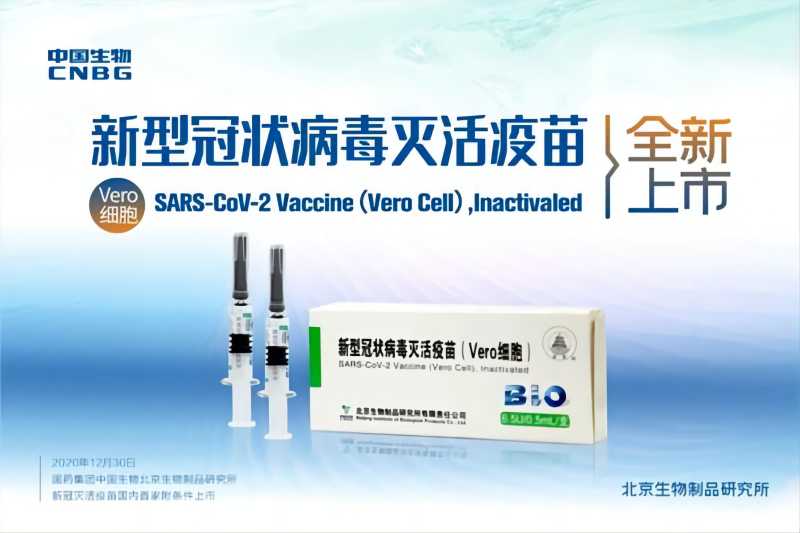
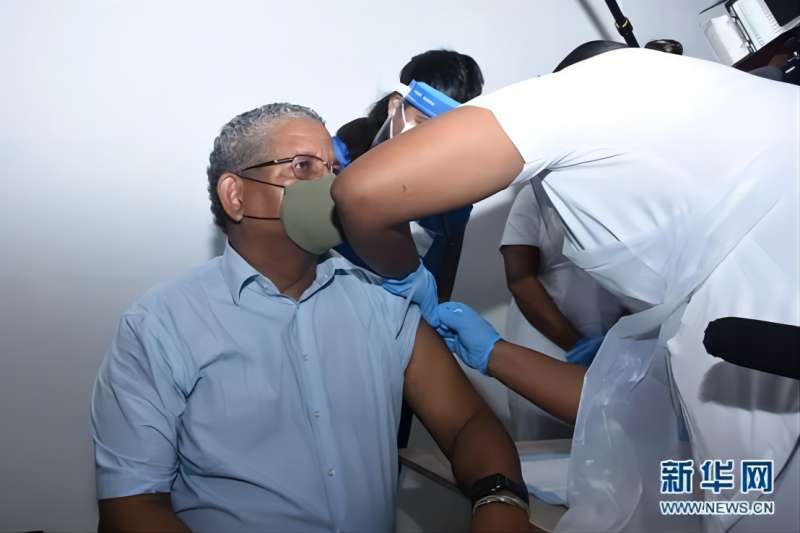
Significant Progress! WHO Extends the Recommended Age of Use of COVID-19 Vaccine (Vero Cell), Inactivated of SINOPHARM CNBG Beijing Institute of Biological Products Co., Ltd. to Over 60 Years Old
Recently, WHO has extended the recommended age of use of COVID-19 Vaccine (Vero Cell), Inactivated developed and produced by SINOPHARM CNBG Beijing Institute of Biological Products Co., Ltd. (BIBP) to over 60 years old, which means that the safety and efficacy of the vaccine for people aged 18 and over have been recognized by WHO. ▲The recommended age o...
View More +Glad Tidings! Production Transformation Model of the Year of Dingge Award 2022
Recently, the much-anticipated “2022 Dingge Award—Digital Transformation Pioneer List” was announced and Beijing Institute of Biological Products (BIBP) subordinate to China National Biotec Group (CNBG) of China National Pharmaceutical Group (SINOPHARM) was awarded the “Production Transformation Model of the Year” for its intelligent upgrade of COVID-19 vaccine filling and ...
View More +Big News!!! BIBP’s Inactivated Vaccine for Omicron Approved by UAE for Clinical Trial
On April 29, theOmicron COVID-19 Vaccine (Vero Cell), Inactivated developed by Beijing Institute of Biological Products Co., Ltd. (BIBP), China National Biotec Group Company Limited (CNBG), Sinopharmwas granted the clinical trial approval by the Department of Health (DoH) – Abu Dhabi, UAE, marking the important development in the global multi-center ...
View More +BIBP’s COVID-19 Vaccine Approved for Emergency Use in Guyana
Mexico City, February 25 (Xinhua News Agency) - Georgetown News: The Guyana’s food and drug regulator has officially approved the Inactivated COVID-19 Vaccine (Vero Cell) developed by Beijing Institute of Biological Products Co., Ltd. (BIBP), China National Biotec Group Company Limited (CNBG), Sinopharm (BIBP’s COVID-19 Vaccine) for emergency use in Guyana, which...
View More +BIBP’s COVID-19 Vaccine Approved for Use in Children Aged 3 to 11 in Argentina
On October 1 local time, Carla Vizzotti, Argentine Minister of Health, announced that the National Administration of Drugs, Foods and Medical Technology of Argentina (ANMAT) has officially approved the vaccination of children aged 3 to 11 in Argentina with the Inactivated COVID-19 Vaccine (Vero Cell) developed and produced by Beijing Institute of Biological Product...
View More +Malaysia Becomes the Eighth Country that Approves BIBP’s COVID-19 Vaccine for Conditional Registration and Marketing
Noor Hisham Abdullah, Director General of Health Malaysia, announced on July 16 that Malalysia’s National Pharmaceutical Control Bureau(NPCB) had approved the Inactivated COVID-19 Vaccine (Vero Cell) developed and produced by Beijing Institute of Biological Products Co., Ltd. (BIBP), China National Biotec Group Company Limited (CNBG), Sinopharm (hereinafter referred to as ...
View More +Turkmenistan Becomes the Seventh Country that Approves BIBP’s COVID-19 Vaccine for Registration and Marketing
On July 6 local time, Turkmenistan (a Central Asian country) announced that it has approved the application for registration of the Inactivated COVID-19 Vaccine (Vero Cell) developed and produced by Beijing Institute of Biological Products Co., Ltd. (BIBP), China National Biotec Group Company Limited (CNBG), Sinopharm (hereinafter referred to as “BIBP’s COVID-19 vaccine”),...
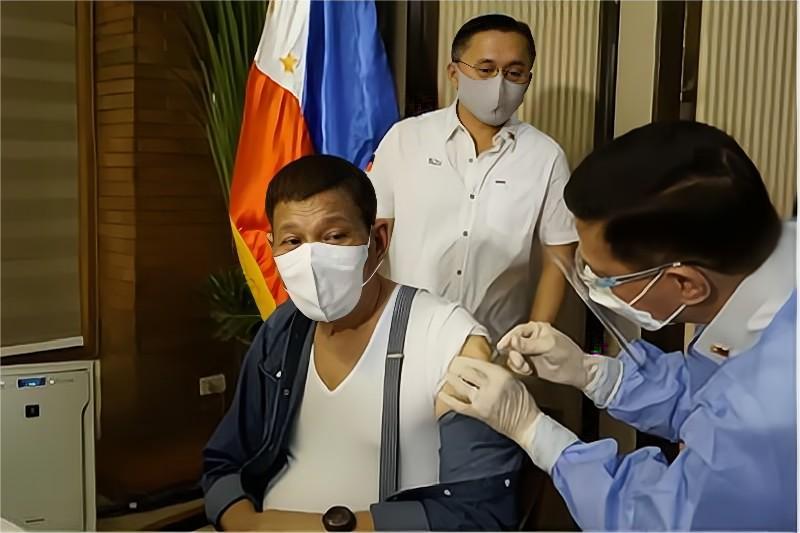
View More +BIBP’s COVID-19 Vaccine Approved for Emergency Use in Philippines
On the evening of June 7 local time, Rolando Enrique Domingo, Director-general of Food and Drug Administration (FDA) of the Philippines said that the Philippines has officially approved the Inactivated COVID-19 Vaccine (Vero Cell) developed by Beijing Institute of Biological Products Anti-COVID-19 Task Force of the Philippine government, which made BIBP’s COVID-19 Vacc...
View More +BIBP’s COVID-19 Vaccine Approved for Emergency Use in Vietnam!
According to the latest news of the Ministry of Health (MOH approved the decision of putting the Inactivated COVID-19 Vaccine (Vero Cell) developed by Beijing Institute of Biological Products Company Limited (CNBG), Sinopharm (hereinafter referred to as “BIBP’s COVID-19 vaccine”) into emergency use, so as to meet the urgent need for the prevention and control of C...
View More +Thailand Becomes the Sixth Country that Approves BIBP’s COVID-19 Vaccine for Registration and Marketing
On May 28 local time, Paisan Dankhum, Secretary General of the Food and Drug Administration (FDA) of Thailand, announced at a press conference that FDA had approved the application for registration of the Inactivated COVID-19 Vaccine (Vero Cell) developed and produced by Beijing Institute of Biological Products Co., Ltd. (BIBP), China National Biotec Group Company Limited ...
View More +BIBP’s COVID-19 Vaccine Granted EUA by WHO for the First Time!
On May 7, Tedros Adhanom Ghebreyesus, Director-general of the World Health Organization (WHO), announced that the Inactivated COVID-19 Vaccine (Vero Cell) developed by Beijing Institute of Biological Products Co., Ltd. (BIBP), China National Biotec Group Company Limited (CNBG), Sinopharm (hereinafter referred to as “BIBP’s COVID-19 Vaccine”) was granted the Emergency U...
View More +China’s COVID-19 Vaccine Obtained EU GMP Certification for the First Time
On April 1, Hungary’s drug regulator issued the EU GMP certification to the Inactivated COVID-19 Vaccine (Vero Cell) developed by Beijing Institute of Biological Products Co., Ltd. (BIBP), China National Biotec Group Company Limited (CNBG), Sinopharm (BIBP’s COVID-19 Vaccine). This is the first vaccine product approved for use and granted GMP certifi...
View More +BIBP’s COVID-19 Vaccine Approved for Emergency Use in Sri Lanka
In the evening of March 19 local time, Channa Jayasumana, State Minister of Production, Supply and Regulation of Pharmaceuticals of Sri Lanka, announced that the National Medicines Regulatory Authority had granted emergency use approval to the Inactivated COVID-19 Vaccine (Vero Cell) developed by Beijing Institute of Biological Products Company Limited (CNBG), Si...
View More +BIBP’s COVID-19 Vaccine Approved for Emergency Use in Lebanon
March 2 (www.haiwainet.cn)- On Monday (March 1) local time, a scientific committee set up by Hamad Hassan, Minister of Public Health of Lebanon Caretaker Cabinet, approved the Inactivated COVID-19 Vaccine (Vero Cell) developed by Beijing Institute of Biological Products Co., Ltd. (BIBP), China National Biotec GroupCompany Limited(CNBG), Sinopharm (BIBP’s COVID-19...
View More +BIBP’s COVID-19 Vaccine Approved for Emergency Use in Nepal
On February 17 local time, the Department of Drug Administration of Nepal issued a notice to approve the Inactivated COVID-19 Vaccine (Vero Cell) developed by Beijing Institute of Biological Products Co., Ltd.(BIBP), China National Biotec Group Company Limited (CNBG), Sinopharm (BIBP’s COVID-19 Vaccine) for emergency use. According t...
View More +Seychelles Becomes the Fourth Country that Approves BIBP’s COVID-19 Vaccine for Emergency Use
On February 25, 2021, the Ministry of Health (MOH) of Seychelles officially approved the Inactivated COVID-19 Vaccine (Vero Cell) developed by Beijing Institute of Biological Products Co., Ltd. (BIBP), China National Biotec Group Company Limited (CNBG), Sinopharm (BIBP’s COVID-19 Vaccine) for registration and marketing in Seychelles, making Seychelles the fou...
View More +